
Stroke and dementia
The ethos of the Geoffrey Jefferson Brain Research Centre is to improve the quality of life for stroke survivors and improve clinical care pathways.
Stroke is a leading cause of death and disability, with around 100,000 people in the UK having a stroke each year, meaning a person has a stroke approximately every five minutes.
There are also currently around 1.3 million stroke survivors living in the UK, requiring a greater focus on rehabilitation and reduction of long-term disability. Learn more about our research within rehabilitation and our focus on reducing long term disability.
Survival rates following a stroke have improved. The national focus looks at how stroke can be prevented, treated quickly and how people can be supported following a stroke.
Stroke is also a risk factor for vascular dementia and depression, and the risk of cognitive impairment is doubled post stroke. Currently, there are very limited treatments for both stroke and dementia, highlighting a huge unmet need within this area.
Our research
Our research covers both ischaemic and haemorrhagic stroke, as well as vascular dementia. Our research also looks to cover the whole translational pathway, from basic science to clinical care and further into community care. We aim to understand:
- the basic biology behind stroke and dementia;
- how to minimise brain damage post stroke;
- how to facilitate repair and recovery of brain function and improved clinical outcomes;
- managing long-term disability with rehabilitation.
Stroke research
Clinical and pre-clinical research
Targeting interleukin-1 for the treatment of acute brain injury
Funder: Brain Research UK
Investigators: Stuart Allan, Craig Smith, Ioana Emilia-Mosneag
Research in the Brain Inflammation Group and by others has shown that there is inflammation in the brain after stroke, and that this inflammation is important in the death of brain cells. It is possible, therefore, that drugs that reduce inflammation could limit brain damage after stroke, representing a possible new treatment option.
Over many years we have investigated this using a drug called anakinra, which works by stopping the action of the protein molecule interleukin-1 (IL-1), which we know can cause inflammation. Anakinra reduces the amount of brain damage in animals with experimental stroke, leading to better recovery of the animals. It is already used in the treatment of other diseases in humans and has been tested in small numbers of stroke patients, with both clots and bleeds. These human trials confirmed that anakinra can reduce inflammation.
However, evidence from one trial suggested that anakinra and tPA might not work well when given together. Testing in animals, the team discovered potential negative interactions between the two drugs, reducing beneficial effects.
In this project, we aim to address two key research questions: firstly whether it is better to give anakinra before rather than after the tPA is injected; and secondly, whether another anti-inflammatory drug (isunakinra) works better than anakinra in reducing brain damage after stroke, and does not have the same problems when given with tPA.
To address these questions, we will use well-established mouse models of stroke in which we can mimic the effects of stroke and test the effects of different treatments by using established measures of brain damage.
DIAGnosis using NOvel technology for Subtypes In Stroke (DIAGNOSIS study)
Stroke caused by bleeding in the brain is known as intracerebral haemorrhage, or ICH for short. ICH is a major cause of death and disability. When ICH patients are in the ambulance on their way to hospital, it may be a vital opportunity to start treatments to prevent further bleeding. Reversing blood thinners and rapid blood pressure lowering are two treatments that we deliver in hospital. Treatment in the ambulance may make them even more effective. However, we currently have no accurate and cost-effective way to tell which patients have an ICH and which have an ischaemic stroke, where a blood clot has blocked an artery.
We have shown [neurologyopen.bmj.com] that simple clinical features such as high blood pressure can be used to identify ICH, but this approach only picks up around half of cases. Therefore, new technologies are needed to improve diagnosis. Our ‘DIAGNOSIS’ study aims to develop an accurate, quick, pragmatic, and cost-effective tool to identify intracerebral haemorrhage in the ambulance. We will recruit up to 300 suspected stroke patients as they arrive in the Emergency Department. We will collect clinical features and test for a marker called GFAP on a rapid lateral flow test. We will combine this test with clinical features to see if they are accurate enough when combined. If so, we aim to then move on to testing accuracy in the ambulance in a future study.
CLArithromycin for Stroke associated Pneumonia (CLASP)
Funder: NIHR
Funding amount: £3.23 million
Pneumonia (a serious infection of the lungs) occurs frequently after a stroke and leads to worse outcomes for patients. Despite modern stroke unit care and antibiotics, the chances of death are three times higher in stroke patients that develop pneumonia, compared to those that don’t. Pneumonia causes excessive, harmful inflammation in the lungs and bloodstream, which contributes to worse outcomes.
Clarithromycin is an antibiotic with anti-inflammatory properties. A team led by Professor Craig Smith (Chief Investigator) will undertake a phase 3, multi-centre, randomised trial of clarithromycin treatment for stroke patients developing pneumonia. The CLArithromycin for post-Stroke Pneumonia (CLASP) trial will take part in 45 stroke units across the UK and will enrol 1166 people hospitalised with a stroke complicated by pneumonia. The trial will determine whether addition of clarithromycin for 5 days to the usual antibiotic treatment received will improve clinical and health economic outcomes.
CLASP is currently in the set-up phase and will start to enrol participating patients in early 2025. For more information contact craig.smith-2@manchester.ac.uk
- Learn more about CLASP https://fundingawards.nihr.ac.uk/award/NIHR158678
Inflammatory drivers of Cardio and Cerebro-vascular disease
Current funder: British Heart Foundation (BHF) Centre of Research Excellence
Funding amount: £8 million (total centre funding)
Inflammation contributes to the risk, progression and outcomes of heart disease, stroke and dementia. The Inflammatory drivers of Cardio- and Cerebro-vascular disease is a research theme within the BHF-funded Centre of Research Excellence at the University of Manchester, co-led by Professor Maya Buch and Professor Craig Smith.
This research funding will further develop our translational “bench to bedside” research capacity with the aim of identifying inflammatory mechanisms that contribute to the risk of vascular disease and dementia, and subsequent clinical outcomes. The ultimate goal of the research theme is to identify and test potential new treatments to reduce the suffering and burden of heart disease, stroke and dementia.
- Learn more at https://www.bhf-cre.manchester.ac.uk/
Doctoral Training Centre for Vascular and Immune contributors to dementia
Funder: Alzheimer’s Society
Funding amount: £2.95 million
In partnership with the Alzheimer’s Society, the Centre is led by Professor Stuart Allan.
We know that blood vessels and changes to the immune system play a key role in the underlying causes of dementia and this centre aims to deepen our understanding of the mechanisms involved and how they could be targeted to develop through new treatment.
By building on this understanding, the students and their supervisors hope to improve diagnosis by identifying the very earliest signs, find ways to modify these signs through new and innovative treatments as well as improve care for patients with dementia.
Visit the Alzheimer’s Society Centre page for more information.
SC IL-1 RA in SAH
Current funder: NIHR EME
This NIHR funded Phase 3 study looked to see if a drug called Kineret, aimed at reducing cytokine (inflammatory molecules) levels in the body improved outcome after a subarachnoid haemorrhage (SAH). SAH is a spontaneous bleed into the brain from an underlying arterial weakness called aneurysm. The study recruited over 600 patients with aneurysmal subarachnoid haemorrhage over a period of 6 years. Outcome was measured at 6 months from treatment completion and final results were available in September 2024. The outcome result was that the drug neither improved nor worsened the outcome of SAH. Whilst the neutral result was disappointing, it has definitively clarified the answer to an important question, based on research in Manchester over the last 20 years.
- Learn more about SC IL-1 RA in SAH.
Stroke-IMPaCT
Current funder: Leducq Foundation, NIH
Stroke-IMPaCT is a network of European and North American researchers working together to discover how inflammation and immune responses contribute to post-stroke cognitive decline.
We want to help develop treatments that completely stop or reduce a person’s risk of developing dementia after a stroke.
- Learn more about Stroke-IMPaCT.
Evaluating the impact of spreading depolarisations in the awake brain
Evaluating the impact of spreading depolarisations, post stroke, in the awake brain using graphene-enabled nanotechnology
Funder: Medical Research Council (MRC)
Funding amount: £1.3 million
Local researchers: Dr Rob Wykes, Professor Stuart Allan and Professor Kostas Kostarelos
Waves of pathological brain signals called spreading depolarisations (SDs) are known to worsen stroke outcome. SDs spontaneously occur post stroke and spread through the brain.
As SDs propagate, they depolarise brain cells, resulting in almost complete ion homeostasis failure. SDs can further reduce cerebral blood flow in areas of the brain that are already experiencing restricted blood flow, and recovery from SD is extremely energy demanding.
A key problem when conducting research to design therapeutic strategies to suppress SDs is the lack of pre-clinical electrophysiological technology capable of detecting and mapping SDs with high spatiotemporal resolution.
The Wykes lab has successfully collaborated with partners on the EU Graphene Flagship, specifically the material scientists and electrical engineers at research institutes in Barcelona (ICN2 and CNM-CISC), who designed and fabricate arrays of graphene-based neurophysiological probes.
We have demonstrated that they are capable of recording SDs across large areas of brain, and are useful for studying pathological brain signals in intact brain. We now aim to bring this cutting-edge technology to preclinical stroke research to gain a better understanding of the mechanisms of SD initiation, and their involvement in worsening stroke severity.
This work will be a crucial first step in the maturity of this technology towards future clinical translation, where we anticipate that it will greatly facilitate management of patients in the neuro-intensive care units.
Furthermore, we aim to design a liposomal-based advanced therapeutic strategy that suppresses SDs to reduce stroke core expansion.
Acute Bundle of Care for Intracerebral Haemorrhage (ABC-ICH)
Previous funders: Health Foundation, Connected Health Cities
Current funders: Health Innovation Manchester, Stroke Association, Innovation Agency, Yorkshire & Humber AHSN, AHSN for the North East and North Cumbria
Local researchers: Adrian Parry-Jones, Hiren Patel, Matt Sutton, Paul Wilson, Lisa Brunton, Emma McManus, Kate Woodward-Nutt, Khalil Kawafi, Appu Suman
External researchers: Martin James (SSNAP), Karla Hemming (University of Birmingham)
The ABC-ICH project was designed to improve survival and recovery from bleeding in the brain or intracerebral haemorrhage (ICH), improving the delivery of acute care for all ICH patients using the ABC care bundle. This was implemented at Salford in 2015-16 and reduced 30-day deaths by a third, saving around 29 lives a year.
Research identified what worked well and what barriers were encountered, refining our approach for the next phase. To provide clinicians with key information at the bedside and allow simple, real-time capture of key performance indicators, we developed the clinician-facing ABC-ICH app and dashboard.
A phased scale-up of the ABC-ICH project across the north of England commenced in 2021, covering 25 hospitals and a population of around 10 million. A Stroke Association Project Grant (2021-24) will allow us to determine whether long-term disability is reduced.
Learn more about ABC-ICH:
- Can a new treatment package improve survival and life after stroke caused by a bleed in the brain? (Stroke Association)
- Scale-up of ABC care bundle for intracerebral haemorrhage across two hyperacute stroke units in one region in England (BMJ Open)
- Video: Patient story – Ann Bamford (YouTube)
Chlorhexidine or toothpaste, manual or powered brushing to prevent pneumonia complicating stroke (CHOSEN)
Current funder: NIHR
Poor oral health is associated with the risk of developing pneumonia after stroke. People with swallowing problems (dysphagia) after stroke are at particular risk. The CHOSEN trial investigated whether oral healthcare treatments, following stroke reduces the likelihood of stroke associated pneumonia (SAP).
Recruitment of participants to the feasibility trial completed in January 2023, with final follow-up in April 2023. We found that the oral healthcare treatments, supported by staff education and training, were feasible, acceptable and well-tolerated. After analysing our results, we plan to undertake a much larger trial across the UK to test cost-effectiveness of the oral healthcare intervention.
- Learn more about CHOSEN.
Assessment of Brain-injury using Radio-Frequency Induction and Microwave Spectroscopy (ABRIMS)
Funder: Engineering and Physical Sciences Research Council (EPSRC) (UK)
Investigator: Adrian Parry-Jones
Stroke is a major cause of death and disability worldwide. Treatments are time critical. The sooner they are given, the better the outcome for patients.
Stroke is often suspected in the ambulance, but up to 40% of these patients turn out to have something else. It is also currently not possible to distinguish between a stroke caused by a blocked artery or one caused by bleeding into the brain. This is important as the treatments are very different.
Furthermore, some treatments can only be given in specialised hospitals, and it is not possible to know if patients need these treatments until they have a brain scan in hospital. If we could confirm a stroke and tell the type of stroke it was in the ambulance, patients could go to the right hospital first time, and treatment could even be started in the ambulance. This will lead to better outcomes in the future for most patients.
This proposal takes advantage of recent work on methods called radio-frequency induction and microwave spectroscopy. We will combine both methods in a single portable device. Our hope is that these techniques will tell us about the changes taking place inside the skull in patients shortly after a stroke. If they do, a portable device for use in ambulances could be developed and further tested, to see if it can improve the speed and access to stroke treatments.
We will test a prototype device to see whether it can distinguish changes in the brain after stroke, comparing heathy volunteers with stroke patients.
- Learn more about the ABRIMS study.
ADAMTS13 (developing novel clot-busting drugs for ischaemic stroke)
We are working on a new thrombolytic or clot-busting drug called ADAMTS13, which shows promising results in animal models of stroke.
ADAMTS13 specifically targets a protein known as von Willebrand factor (VWF), which contributes to clot formation and is common in clots seen in patients with ischaemic stroke.
We have developed a variant of this protein that is constantly active – caADAMTS13 – which enables it to break down even more VWF than the native protein.
Recent research in our group has shown that caADAMTS13 is better at breaking down platelet-rich clots than the current thrombolytic drugs licensed for use in stroke patients, and may reduce the risk of bleeding associated with current treatments.
We are now scaling up this research to optimise the dose of caADAMTS13 that can be given and to test if caADAMTS13 can be safely given over a longer time window, which would mean more patients could be treated with it if it was eventually licensed. In addition, using blood clots retrieved from stroke patients by thrombectomy, we are testing how good caADAMTS13 is at breaking down these clots, compared to the current standard treatment rtPA.
Excitingly, we hope to see this drug in clinical trials within the next five years.
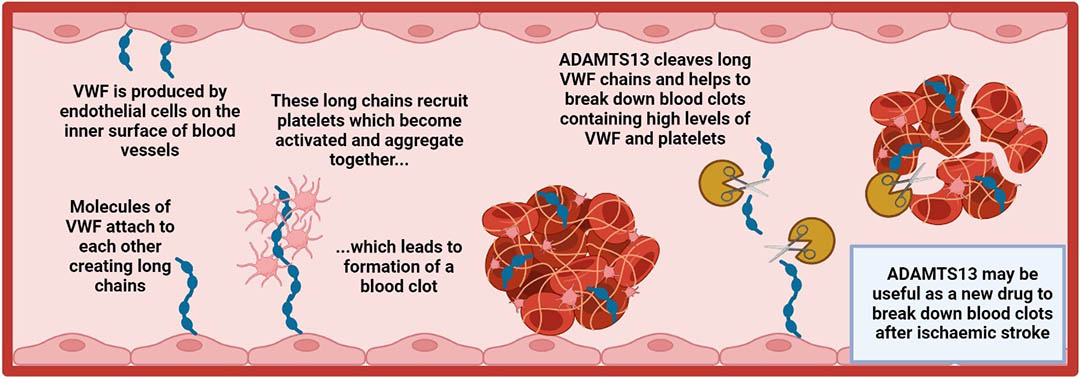
The illustration above shows how blood clots form, and how ADAMTS13 could break them down.
VWF is produced by endothelial cells on the inner surface of blood vessels.
Molecules of VWF attach to each other, creating long chains.
These long chains recruit platelets, which become activated and aggregate together… which leads to the formation of a blood clot.
ADAMTS13 cleaves long VWF chains and helps to break down blood clots containing high levels of VWF and platelets.
ADAMTS13 may be useful as a new drug to break down blood clots after ischaemic stroke.
Modulating cholesterol 25-hydroxylase
Modulating cholesterol 25-hydroxylase: a strategy to accelerate haematoma clearance after intracerebral haemorrhage to improve outcome
Funder: Medical Research Council
Funding amount: £757,669.63
Duration: 3 years (October 2023 to October 2026)
Manchester investigators: Catherine Lawrence (PI), Paul Kasher, Stuart Allan, Ben Dickie
Intracerebral haemorrhage is a type of stroke that occurs when a blood vessel suddenly ruptures and begins bleeding into the brain. Blood that is now in the brain contains various cells (including red blood cells) that start to release ‘toxic factors’ that can cause further damage to brain cells. It is important therefore, to try and get rid of the blood as quickly as possible, to reduce the chances of it causing any harm.
Our tissues/organs contain a group of cells called ‘phagocytes’ whose job is to eat any harmful cells and smaller particles to try to protect our bodies from any of their harmful effects. In haemorrhagic stroke, these ‘phagocytes’ try to eat up the red blood cells and toxic factors. However, this job can take time and trying to find a way to help the phagocytes get rid of the blood quicker and more efficiently would help to prevent further damage to the brain.
We have discovered that the amount of a protein called ‘cholesterol 25-hydroxylase’ (Ch25h) goes up in the phagocytes after haemorrhage, and we believe that this protein might help the phagocytes to work better to clear away the blood. We will bring together a unique team of scientists who will use animal models of brain haemorrhage, as well as tissue from patients who died from this condition to learn more about what Ch25h is doing in the brain.
We will try to work out if Ch25h can help the phagocytes to remove blood. Normally in cells, Ch25h stimulates the production and release of other molecules including 25-hydroxycholesterol (25-HC). We will therefore give 25-HC to animals that have brain haemorrhage and test if this increases how well the blood is removed from the brain, and if it can also improve the symptoms seen in this type of stroke.
By doing this work, we hope to identify a potential new way to improve outcome and reduce the negative impact of haemorrhagic stroke, a life-threatening condition with no current treatment options.
Kasher Lab
The Kasher Lab is a zebrafish lab with interests in stroke, neuroinflammation, genetics and neurological disease.
Twitter: @KasherLab
Brain Inflammation Group
The Brain Inflammation Group focuses on stroke. It also investigates other cerebrovascular diseases such as Alzheimer’s disease and vascular dementia.
The team works to understand the inflammatory mechanisms in healthy versus diseased brains to identify novel therapeutic targets.
Twitter: @BIG_research
Dementia research
Dementia is the leading cause of death in the UK (ONS 2022). Over 50 million people worldwide live with one of the diseases underlying dementia.
However, novel treatments are on the horizon for several major causes, including Alzheimer’s disease and cerebrovascular disease. To ensure people have access to treatment before disease causes significant damage to neurons, we must make a diagnosis at an early disease stage.
This is best done by examining biomarkers in either blood or cerebrospinal fluid. Together with collaborators, we are preparing the ground for the availability of disease-modifying medications for the diseases that cause dementia.
We’re doing this by conducting clinical trials, rolling out biomarkers, and examining dementia’s links between other morbidities, such as hearing loss. Our researchers work closely with patient and public groups, such as the South Asian community, to shape their work.
Greater Manchester Dementia Research Centre (GMDRC)
The Greater Manchester Dementia Research Centre (GMDRC) is Greater Manchester’s leading clinical trial unit for the diseases causing dementia. It is led by Dr Ross Dunne, our dementia theme lead.
Learn more on the GMDRC website.
Greenstein Lab (vascular dementia research)
 Investigator: Adam Greenstein
Investigator: Adam Greenstein
Blood vessels are the body’s irrigation system, enabling nutrients and oxygen in the blood to be delivered from the top of your head to the end of your toes. It is easy to think of blood vessels as pipes of a fixed size, which supply a set volume of blood to a particular organ.
However, this would lead to swelling of vulnerable organs such as the brain when blood pressure rises. Therefore, when blood pressure does increase, the arteries sense this, and become narrower to make sure blood flow is kept to a constant level and maintain optimum brain health.
In both Alzheimer’s disease and vascular dementia, there is a reduction in blood flow to the brain, which lowers the amount of nutrients and oxygen delivered to the brain. Over a period of time, this leads to loss of cognition.
Our research shows that in models of Alzheimer’s disease and hypertension-driven vascular dementia, the arteries are too sensitive to intraluminal pressure and over constrict to reduce cerebral blood flow.
Our research is identifying the mechanisms that drive this over-constriction and trying to pharmacologically restore the arterial function, helping identify novel treatments for people living with dementia.
- Learn about the Microvascular @ Manchester research group.
All-in-One Study
Funder: Alzheimer’s Society
Local researchers: Laura Parkes, Ross Dunne, Karl Herholz, Oli Sparasci, Alistair Burns
External researchers: John O’Brien, Franklin Aigbirhio (University of Cambridge), Henrik Zetterberg (UCL)
Dementia is the leading cause of death in the UK, and Alzheimer’s underlies 60% of all dementia. There are currently no medications that slow progress available in the UK.
Disease-modifying medications for Alzheimer’s disease have been licensed in the United States and are under scrutiny by NICE. Evidence suggests that the protein abnormalities in Alzheimer’s disease (Beta amyloid and tau) can be detected in the CSF up to ten years before the onset of cognitive symptoms.
Our work is focused on getting research discoveries into clinical practice in the NHS. Our All-in-One Study, funded by the Alzheimer’s Society, will evaluate new brain scanning technologies alongside a blood test for Alzheimer’s disease, to see if we can speed up diagnosis and make It more accurate.
We are also working with our colleagues in The University of Manchester’s Hearing Health Biomedical Research Centre to evaluate the use of hearing tests to predict cognitive decline alongside novel blood biomarkers. While blood tests remain solely for research use, the analysis of cerebrospinal fluid – which bathes the brain – is being rolled out in our Mental Health Trust.
We are working with industry partners Stratastem and other companies to develop platforms for the development of new medications for use in Alzheimer’s and other diseases underlying dementia.
In conjunction with the UK’s Brain Health Collaboration, our novel clinic at brainHealth Manchester uses advanced cognitive testing, fluid biomarkers and neuroimaging to diagnose the earliest stages of Alzheimer’s and other neurodegenerative diseases, so that people can participate in research, and when the time comes, have disease-modifying medications to slow their progress.
- Learn more about the All-in-One study.
Investigators
 Craig Smith
Craig Smith
Theme lead
Read Craig’s research profile
 Ross Dunne
Ross Dunne
Theme lead
Read Ross’ research profile
 Adrian Parry-Jones
Adrian Parry-Jones
 Hiren Patel
Hiren Patel
 Nigel Hooper
Nigel Hooper
 Audrey Bowen
Audrey Bowen
 Tao Wang
Tao Wang
 Kieron South
Kieron South
 Stuart Allan
Stuart Allan
 Adam Greenstein
Adam Greenstein
 Paul Kasher
Paul Kasher
 Cath Lawrence
Cath Lawrence
 Emmanuel Pinteaux
Emmanuel Pinteaux
 Ingo Schiessl
Ingo Schiessl
 Sarah Ryan
Sarah Ryan
 Harry Pritchard
Harry Pritchard
 Doug Dyer
Doug Dyer
 Roy Chun-Laam Ng
Roy Chun-Laam Ng Rob Wykes
Rob Wykes David Brough
David Brough