Study shows safety of world’s first human graphene-based brain computer interface operation
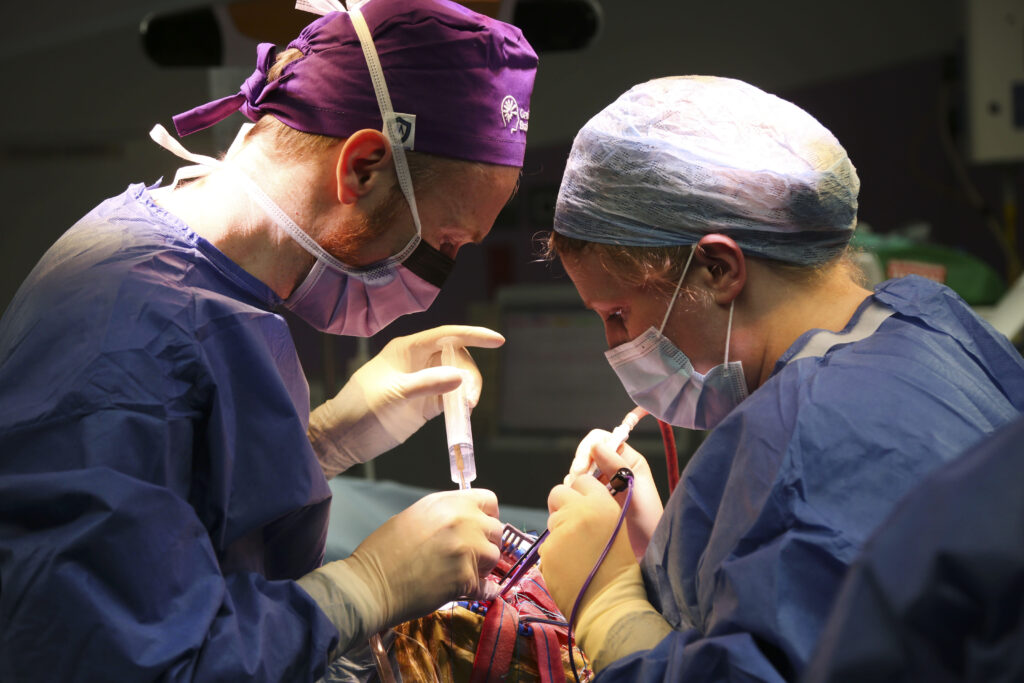
Eight patients have been treated at Northern Care Alliance (NCA) in the world’s first-in-human study of graphene-brain interfaces to map and monitor brain activity and so improve precision during brain tumour surgery.
The study, sponsored by The University of Manchester and conducted with NCA, evaluated INBRAIN Neuroelectronics’ graphene-based cortical interface device. It showed a favourable perioperative safety profile with no device-related adverse events observed in all eight patients treated up to surgical discharge.
In the study, INBRAIN’s graphene electrodes were used alongside standard-of-care monitoring systems during tumour surgery. In select cases involving awake surgery, patients performed tasks such as naming objects, enabling researchers to evaluate the system’s performance to decode speech representation in the brain in high resolution.
 “The ability to detect high-frequency neural activity with micrometer-scale precision and also modulate it provides a fundamentally new level of insight into brain–tumor interactions and functional brain decoding and mapping. This level of resolution has the potential to significantly improve surgical precision and open new avenues for treating neurological disorders,” said Dr David Coope, chief clinical investigator and consultant neurosurgeon at the Manchester Centre for Clinical Neurosciences (part of NCA), and the Geoffrey Jefferson Brain Research Centre, a partnership between NCA and The University of Manchester.
“The ability to detect high-frequency neural activity with micrometer-scale precision and also modulate it provides a fundamentally new level of insight into brain–tumor interactions and functional brain decoding and mapping. This level of resolution has the potential to significantly improve surgical precision and open new avenues for treating neurological disorders,” said Dr David Coope, chief clinical investigator and consultant neurosurgeon at the Manchester Centre for Clinical Neurosciences (part of NCA), and the Geoffrey Jefferson Brain Research Centre, a partnership between NCA and The University of Manchester.
During brain surgery, graphene electrodes are used to map and monitor neural activity, helping surgeons identify and preserve critical functions such as movement and speech. However, conventional electrodes are limited by their rigidity, size, and signal sensitivity, which can restrict their ability to conform to the brain’s complex surface and capture detailed neural activity.
The graphene-based electrodes represent a new class of neural interface technology designed to overcome these challenges. Ultra-thin, micrometric and highly flexible, they closely adapt to the brain’s contours and access hard-to-reach areas. By replacing metal contacts with graphene, a highly sensitive carbon-based material, these devices enable higher-resolution signal detection and more precise stimulation, supporting real-time, more detailed brain decoding and mapping during surgery.
 “This study demonstrates that graphene can safely interface with the human brain, and capture neural signals with exceptional fidelity and resolution to enable precise decoding of brain and speech-related patterns metals can barely see,” said Dr Kostas Kostarelos, co-founder of INBRAIN Neuroelectronics and chief scientific investigator of the study. “It marks a pivotal step towards translating a new enabling technology using neural signals into meaningful clinical applications and real-world patient benefit.”
“This study demonstrates that graphene can safely interface with the human brain, and capture neural signals with exceptional fidelity and resolution to enable precise decoding of brain and speech-related patterns metals can barely see,” said Dr Kostas Kostarelos, co-founder of INBRAIN Neuroelectronics and chief scientific investigator of the study. “It marks a pivotal step towards translating a new enabling technology using neural signals into meaningful clinical applications and real-world patient benefit.”
Carolina Aguilar, CEO and Co-founder, INBRAIN Neuroelectronics said: “Graphene has the potential to fundamentally change how we interface with the brain, enabling higher resolution of neural function specific biomarkers, safer, and more intelligent BCI systems. We look forward to announcing the full results this year, as we advance towards commercialization.”
 About INBRAIN Neuroelectronics
About INBRAIN Neuroelectronics
INBRAIN Neuroelectronics is a clinical-stage neurotechnology company developing graphene-based brain-computer interface therapeutics. The company’s platform leverages the unique properties of graphene to create high-resolution, minimally invasive neural interfaces designed to treat neurological disorders in central and peripheral nervous system applications powered by AI. INBRAIN is advancing a pipeline of BCI-based therapies aimed at decoding and modulating neural activity with unprecedented precision and intelligence. For more information, visit www.inbrain-neuroelectronics.com.



0 Comments